Thermal Energy
What is Thermal Energy? Thermal Energy is the total Potential and Kinetic energy of all the particles in an object. Thermal energy depends upon the mass, temperature, and phase of an object.
Two substances can be the same temperature and have different thermal energies.
Two substances can be the same temperature and have different thermal energies.

The thermal energy depends on mass and temperature. The tea is at a higher temperature than the lemonade because its particles have a higher average kinetic energy. The lemonade is at a lower temperature, but it has more thermal energy because it has many more particles.
Thermal Expansion and Contraction
Thermal contraction occurs when matter is heated, because particles of matter tend to move closer together as temperature increases.
Thermal expansion occurs because particles of matter tend to move farther apart as temperature increases. Gassed expand more than liquids and liquids usually expand more than solids. A gas expands more easily than a liquid or solid because the forces of attraction among particles in a gas are weaker.
Thermal expansion occurs because particles of matter tend to move farther apart as temperature increases. Gassed expand more than liquids and liquids usually expand more than solids. A gas expands more easily than a liquid or solid because the forces of attraction among particles in a gas are weaker.
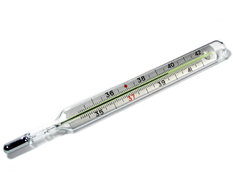
Thermal expansion is used in glass thermometers. As the temperature increases, the alcohol in the tube expands and its height increases. The increase in height is proportional to the increase in temperature.
Work and Heat
Heat is the transfer of thermal energy from on object to another as the result of a difference in temperature. Heat flows spontaneously from hot objects to cold objects.
Specific Heat
The amount of heat needed to raise the temperature of one gram of material by one degree Celsius is a calorie. If equal masses of iron and plastic absorb the same heat, the iron temperature rises more. The lower a material's specific heat, the more its temperature increases when heat is absorbed. Heat is measured in Joules or calories. One calorie is the energy needed to raise the temperature of one gram of water by one degrees Celsius. When a car is heated by the sun, the temperature of the plastic bumper. The iron in the door has a lower specific heat than needed to raise temperature of one gram of a material by one degree.
Measuring Heat Changes
A Calorimeter is used to measure changes in thermal energy. A Calorimeter uses the principle that heat flows from a hotter object to a colder object until both reach the same temperature. According to the law of conservation of energy, the thermal energy released by a test sample is equal to the thermal energy absorbed by its surroundings. The calorimeter is sealed to prevent thermal energy from escaping.
Temperature
Temperature is the measure of how hot or cold an object is compared to reference point. On the Celsius scale, the reference points for temperature are the freezing and boiling points of water. On the Kelvin scale, another reference point is absolute zero, which is defined as a temperature of 0 kelvins. Temperature is related to the average kinetic energy of the particles in an object due to their random motions through space.
When an object heats up its particles move faster, on average.
When an object heats up its particles move faster, on average.